In Alzheimer’s disease(AD), the pathophysiological changes of brain Aβ and Tau occur 15 to 20 years before the onset of symptoms. If the pathophysiological changes can be detected in time before symptoms appear, and corresponding intervention measures can be taken, this will help reduce the incidence and prevalence of AD. At present, the research of peripheral markers has become a hot issue in the field of AD. The study of peripheral markers mainly focuses on the study of Aβ and Tau in cerebrospinal fluid(CSF), which should not be widely carried out due to their aggressiveness. As a diagnostic marker of AD, peripheral blood is less traumatic, easy to sample, suitable for extensive development, but has low sensitivity. How to find more sensitive markers in peripheral blood has become a key issue to be solved urgently.
In August 2019, Jianping Jia ‘s team from Xuanwu Hospital published an online paper entitled “Concordance between the Assessment of Aβ42, T-tau, and P-T181-tau in Peripheral Blood Neuronal-derived Exosomes and Cerebrospinal Fluid” in Alzheimer’s & Dementia (IF :14.423) , found in the peripheral blood biomarkers in the diagnosis of AD is equivalent to the effectiveness of the CSF. It will have a major impact on the diagnostic mode AD.

The first phase of the study was the discovery phase, which included subjects from Beijing. And the second phase was the verification phase, which included subjects from Guizhou, Shandong, Henan, Jilin, Inner Mongolia, Guangxi, and Hebei provinces. Research needs to systematically observe and compare the peripheral blood and cerebrospinal fluid of the same subject, which brings great challenges and difficulties to the research. The study used Western Blot, ELASA, co-immunoprecipitation and transmission electron microscopy experiments to do detailed biological verification. The results showed that in the peripheral blood neurogenic exosomes, the levels of Aβ42, T-tau, and P-tau in AD patients were significantly different from those with mild cognitive impairment and controls, and the three could be clearly distinguished. The correlation analysis between peripheral blood neurogenic exosomes and cerebrospinal fluid biomarkers found that the levels of Aβ42, T-tau and P-tau in neurogenic exosomes are highly correlated with their levels in cerebrospinal fluid. It shows that exosomal biomarkers may reflect the pathophysiological changes of the brain, which are equivalent to the diagnostic power of cerebrospinal fluid, and can be used to diagnose AD.
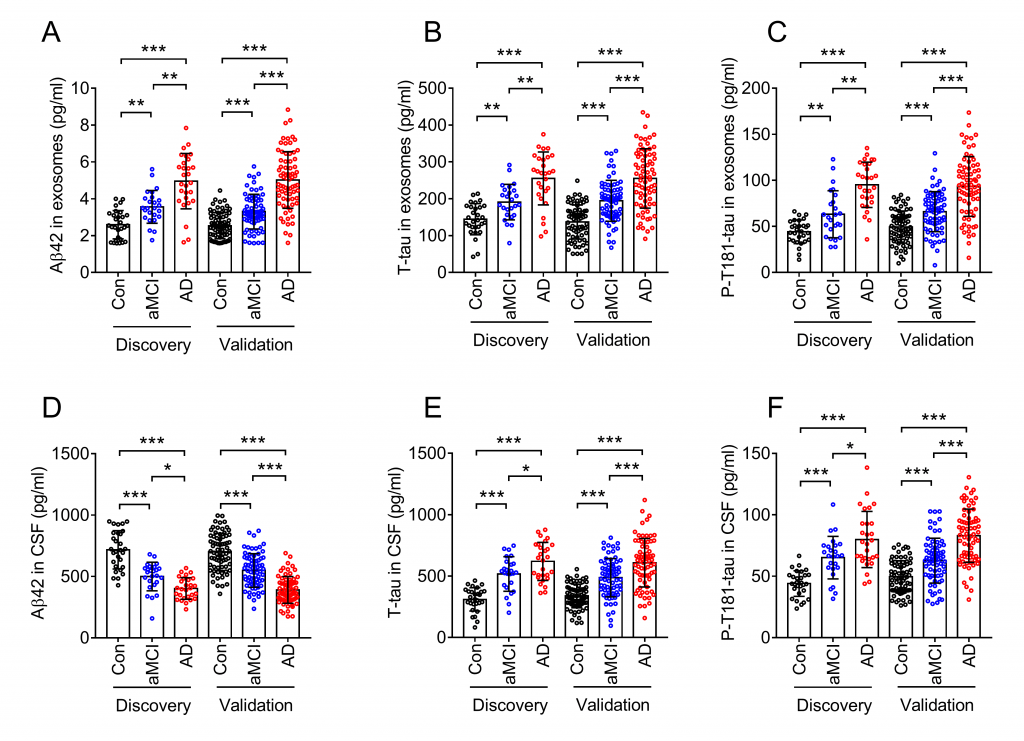
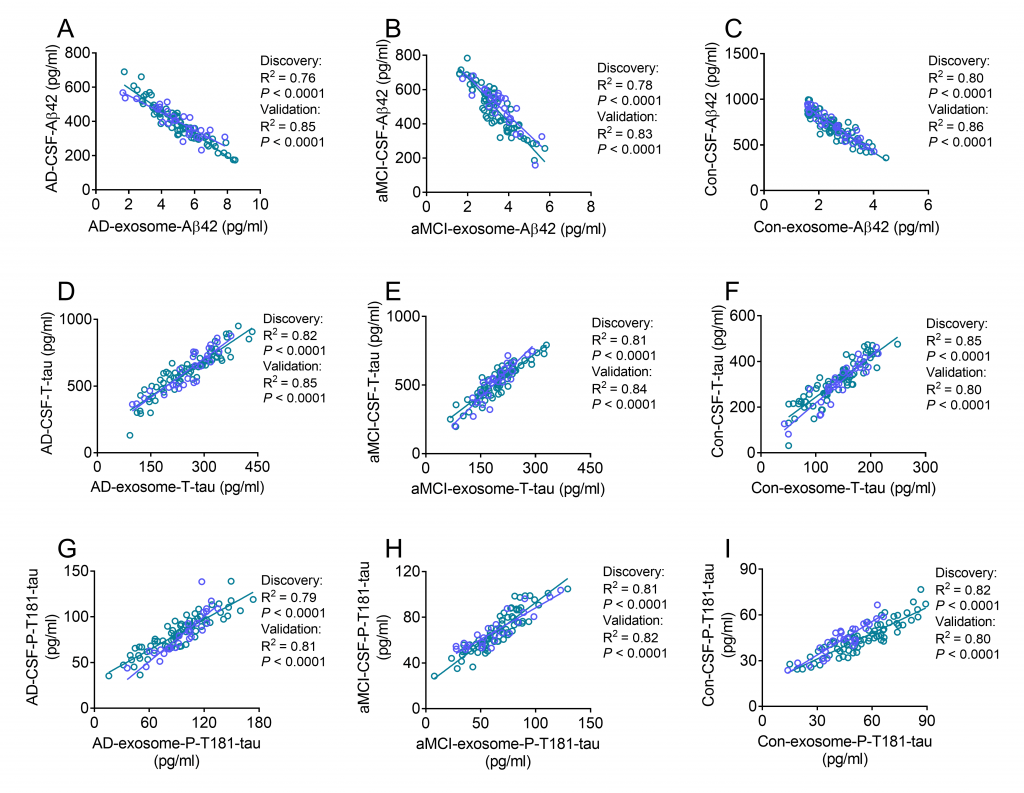
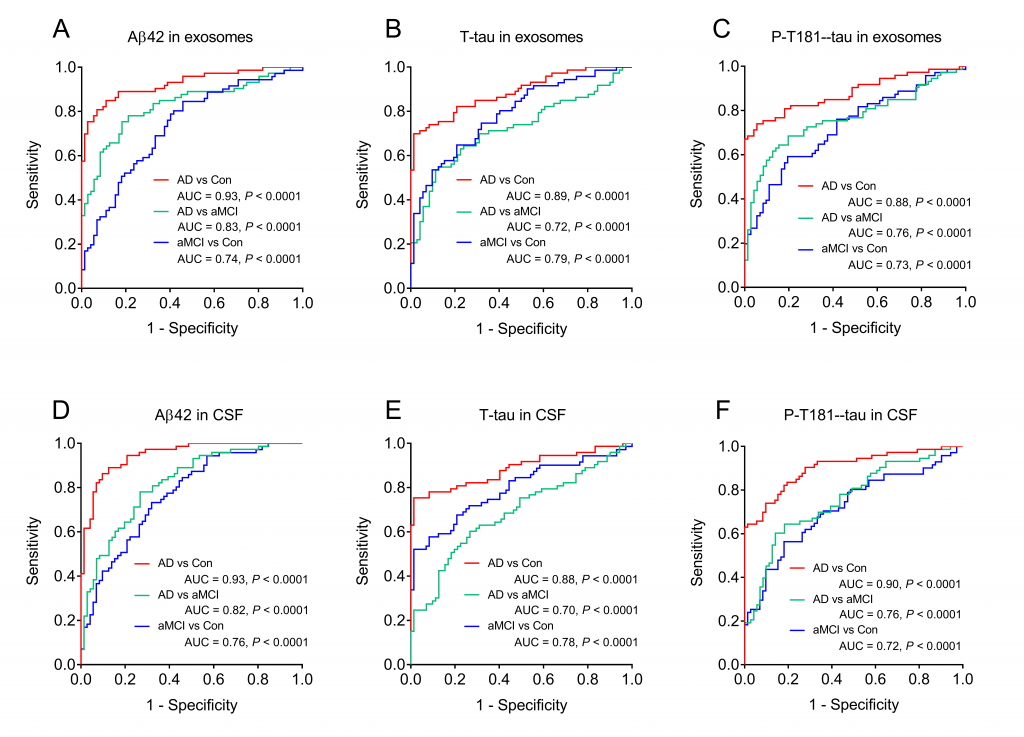
The study also calculated the receiver operating curve (ROC) of Aβ42, T-tau and P-tau in exosomes and cerebrospinal fluid, and the results showed a significantly high area under the curve (AUC). By analyzing and comparing the AUC of AD/mild cognitive impairment, it is found that Aβ42, T-tau and P-tau in peripheral blood neurogenic exosomes and cerebrospinal fluid are powerful evidences for the diagnosis of diseases. The DeLong test found that Aβ42, T-tau and P-tau in neurogenic exosomes have the same diagnostic capabilities as their counterparts in cerebrospinal fluid, providing a new method for the diagnosis of AD. After that, the three composite biomarkers were evaluated through logistic regression analysis, and compared through ROC curve analysis, combined with Aβ42, T-tau and P-tau to establish an AD diagnostic model.
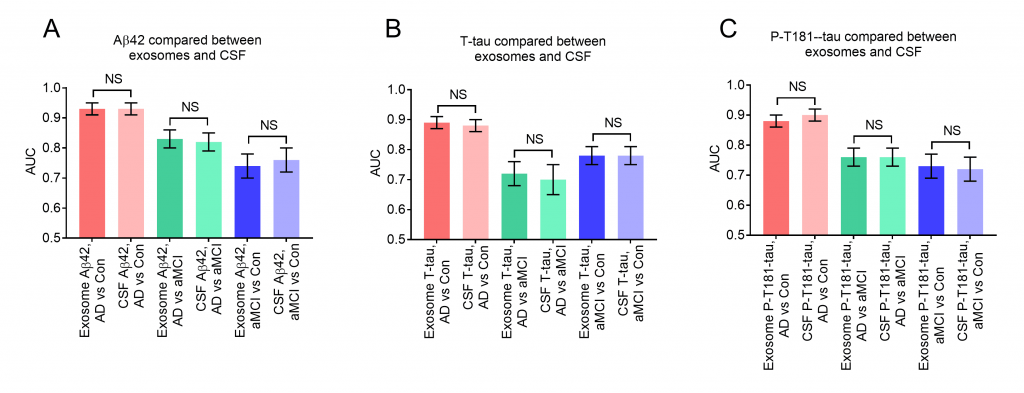
This study compares the diagnostic efficacy of blood and cerebrospinal fluid peripheral markers of the same patient for the first time in the world, and found that the diagnostic efficacy of biomarkers of exosomes in peripheral blood is equivalent to that of cerebrospinal fluid in the diagnosis of AD. For the first time, the voice of the Chinese in the diagnosis of AD peripheral markers was reported, and the research results of the Chinese were reported. This result was published in the most influential journal in the international AD industry, providing for the formulation of precise intervention programs before the onset of AD clinical symptoms An important basis.
This study was supported by the Key Project of the National Natural Science Foundation of China (81530036); the National Key Scientific Instrument and Equipment Development Project (31627803); Mission Program of Beijing Municipal Administration of Hospitals (SML20150801); Beijing Scholars Program; Beijing Brain Initiative from Beijing Municipal Science & Technology Commission (Z161100000216137); CHINA-CANADA Joint Initiative on Alzheimer’s Disease and Related Disorders (81261120571) and Beijing Municipal Commission of Health and Family Planning (PXM2018_026283_000002).
